Article Text
Abstract
Background The resolution of potential drug-related problems is a priority of pharmaceutical care programmes.
Objectives To assess the clinical impact on drug-related negative outcomes of a pharmaceutical care programme focusing on the resolution of potential drug-related problems, initiated in the emergency department for patients with heart failure (HF) and/or chronic obstructive pulmonary disease (COPD).
Methods Controlled trials, in which older adults (≥65 years) receiving four or more medications admitted to the emergency department for ≥12 hours for worsening of HF and/or COPD were randomised (1:1) to either a pharmaceutical care programme focusing on resolving potential drug-related problems initiated at the emergency department (intervention group (IG)) or standard care (control group). Comparisons between the groups were made for the proportion of patients with drug-related negative outcomes, number of drug-related negative outcomes per patient, mean stay, patients readmitted within 180 days and 180-day mortality.
Results 118 patients were included, 59 in each group. Fewer patients in the IG had drug-related negative outcomes (37 (62.7%) vs 47 (79.7%) in the control group (p=0.042)). Fewer drug-related negative outcomes per patient occurred in the IG (56 (0.95 per patient) vs 85 (1.44 per patient) in the control group (p=0.01)). The mean stay was similar between groups (194.7 hours in the IG vs 242.5 hours in the control group (p=0.186)). No difference in revisits within 180 days was found (32 (54.24%) in the IG vs 22 (37.3%) in the control group (p=0.065)). 180-Day mortality was detected in 11 (18.6%) patients in the IG compared with 13 (22%) in the control group (p=0.647).
Conclusion A pharmaceutical care programme focusing on resolving potential drug-related problems initiated at the emergency department has a favourable clinical impact, as it reduces the number and prevalence of drug-related negative outcomes. No difference was found in other outcome variables.
Trial registration number NCT02368548.
- Pharmaceutical care
- Emergency Department
- clinical trial
- Drug-related negative outcomes
- Heart Failure
- COPD
Statistics from Altmetric.com
- Pharmaceutical care
- Emergency Department
- clinical trial
- Drug-related negative outcomes
- Heart Failure
- COPD
What this paper adds
What is already known on this subject
Morbidity related to drug-related negative outcomes (DNO) is a major health concern in Western countries.
Evidence to support the clinical impact of pharmaceutical care programmes focusing on the resolution of potential drug-related problems is unclear.
What this study adds
This study adds knowledge about the impact of pharmaceutical care programmes focusing on the resolution of potential drug-related problems, and has a favourable clinical impact, reducing the number and prevalence of DNO.
Introduction
Populations in more developed countries are ageing rapidly. It is estimated that 34% of the people in Europe will be older than 60 years by 2050.1 Ageing is strongly associated with multiple morbidities and increased use of medications. Polypharmacy, defined as the practice of administering multiple medications over a prolonged period, is considered a risk factor for geriatric syndrome and recognised as linked to increased risk of drug-related negative outcomes (DNO), defined as health problems that patients experience due to drug use or non-use.2
There is strong evidence that morbidity related to DNO is a major health problem in Western countries. In a meta-analysis evaluating 35 prospective studies, Lazarou et al 3 ranked DNO between the fourth and sixth cause of in-hospital death. Up to 38% of emergency department (ED) visits are associated with DNO, of which up to 70% are avoidable.4–6 Furthermore, DNO are directly responsible for 16–19% of hospital admissions in Spain, and over half of these DNO are considered preventable.7
Prevention and resolution of potential drug-related problems are priorities for pharmaceutical care programmes. Several studies have assessed the impact of pharmaceutical care programmes in areas of care other than the ED. In these studies, resolution of potential drug-related problems was associated with a decrease in the prevalence of DNO8 and readmissions.9 10 To the best of our knowledge, the extent to which the resolution of potential drug-related problems may affect clinical outcomes, such as prevalence of DNO, mortality, or the number of readmissions to the ED, is yet to be explored. The few studies focusing on EDs have shown a reduction in the number of potential drug-related problems following the implementation of specific pharmaceutical care programmes.11–13 However, these studies did not evaluate the correlation between the decrease in potential drug-related problems and the occurrence of negative clinical outcomes.
Pharmaceutical care services aiming to resolve potential drug-related problems should prioritise patients with conditions known to have a stronger effect on health outcomes and healthcare expenditure. Heart failure (HF) and chronic obstructive pulmonary disease (COPD) are prevalent chronic conditions related to comorbidity, polypharmacy, DNO and frequent exacerbations.14 15
This study aimed to assess the clinical impact on DNO of a pharmaceutical care programme focusing on the resolution of potential drug-related problems, initiated in the ED for patients with chronic HF and/or COPD.
Methods
Trial design
Randomised controlled trial, which involved patients who were allocated in a 1:1 ratio to participate in a pharmaceutical care programme initiated at the ED centred either on the resolution of potential drug-related problems (intervention group) or administered as standard care (control group). Neither patients nor healthcare professionals were blinded to the treatment group, in accordance with the nature of the intervention.
Participants
Patients who met the following criteria were eligible for inclusion: 65 years or older, length of stay in ED longer than 12 hours, decompensation of HF and/or COPD and polypharmacy (four or more drugs). Institutionalised patients and those with severe cognitive deficits or mental illness documented in the medical record were excluded.
The study protocol was reviewed and approved by the ethics committee of the Hospital de la Santa Creu i Sant Pau. The study procedures complied with the ethical principles set out in the Declaration of Helsinki. All participants were informed about the aims and characteristics of the study and agreed to participate by written informed consent. The study was registered in the ClinicalTrials registry (trial registration number NCT02368548).
Randomisation
Randomisation was performed by the hospital’s pharmacology department using SPSS V.18 (SPSS, Chicago, Illinois, USA) to create a dedicated application to randomise patients to one of the of two study groups (distribution 1:1). The application used a seed obtained by rolling two dice to select the row and column from a random-number table; therefore, while replicable but unpredictable, the series was perfectly balanced between groups in 10-case blocks.
Random patient selection was carried out between 2 January 2012 and 26 February 2013. Assessment of eligibility and ulterior distribution across groups was conducted chronologically by the pharmacist according to the randomisation scheme. The impact of seasonal variations was considered to be minimal owing to the 12-month duration of the study and the randomisation process.
Study setting
The study was conducted in a tertiary referral hospital from Catalonia, Spain, serving about 437 000 inhabitants, with an annual volume of approximately 150 000 ED admissions (including adult/general ED, paediatric ED, psychiatric ED and gynaecology and obstetrics ED), 68 000 adult/general ED admissions and 34 000 hospital admissions.
Intervention
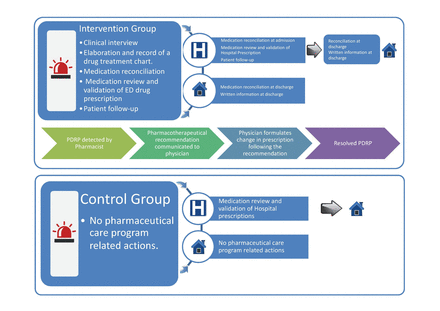
The intervention consisted of a pharmaceutical care programme focusing on the resolution of potential drug-related problems, from admission to ED until discharge (figure 1).
Intervention description. ED, emergency department; PDRP, potential drug-related problems.
Implementation of the programme was exclusively conducted by the same clinical pharmacist for the intervention group. The curricular characteristics of this pharmacist were chosen so that they could be extrapolated to most pharmacists working at an ED in Spain: hospital pharmacist specialist, clinical skills in chronic care management and emergency procedures, demonstrated by 5 years’ experience. The pharmaceutical care programme comprised the following steps:
Obtaining and recording the medication chart. As part of this process, the pharmacist confirmed, by interviewing the patient or caregiver, the medication taken at home as listed in the electronic health records.
Medication reconciliation in each of the care transitions. Medication reconciliation is defined by the Institute for Healthcare Improvement as ‘the process of creating the most accurate list possible of all medications a patient is taking—including drug name, dosage, frequency and route—and comparing that list against the physician’s admission, transfer and/or discharge orders, with the goal of providing correct medications to the patient at all transition points within the hospital’.
Medicine review and validation of physician prescriptions during the stay at the ED and during hospitalisation. This consisted of reviewing the following aspects of the patient’s medication: (a) the indication for each medication in relation to the patient’s condition and (b) the appropriateness of each medication, dose, schedule, duration of the treatment for the patient’s age and/or clinical status (renal function or liver function). In addition, therapeutic drug monitoring was performed for drugs with a narrow therapeutic range.
Patient follow-up. This consisted of evaluation of the effectiveness and safety of the treatment according to standard clinical practice and patients’ objective data from clinical records.
Provision of additional written information at discharge, with clear indications for drug therapy regimen using software tools provided by the Catalan Drug Information Centre (CedimCat).16
When potential drug-related problems were detected, the prescribing physician was informed by means of the electronic health record. The pharmacist reported the problem causing this problem and proposed an alternative prescription that would be available for the health team in the electronic health record.
Potential drug-related problems that, after having been relayed to physicians, generated a change in medical prescription according to the recommendations given by the pharmacist in less than 24 hours after the pharmacist advice were considered resolved potential drug-related problems.
Patients in the control group received standard pharmaceutical care, initiated at admission to the ward and consisting of medication review and prescriptions' validation, analogous to step 3 in the intervention group.
Only resolved potential drug-related problems detected in the pharmaceutical care programme were recorded. Resolved potential drug-related problems were classified according to the Third Consensus of Granada.17 The Anatomical Therapeutic Chemical (ATC) Classification System code of the drugs involved in the resolved potential drug-related problems was also recorded.
Outcomes
Variables likely to modify the effects of medication, such as age, sex, number of medications being taken, number of chronic conditions, hypertension, diabetes mellitus, dyslipidaemia, atrial fibrillation, ischaemic heart disease, renal failure and Charlson index were recorded at admission.
Final outcome variables collated in our study included:
DNO defined as health problems that patients experience owing to drug use or non-use.2 In both groups, DNO were detected by retrospective revision of the electronic health record after discharge. We defined DNO as events specifically pointed out in the electronic health record or any of the following situations previously defined by an independent physician: (a) poor glycaemic control—that is, glucose ≥180 or ≤50 mg/dL in men or ≤45 mg/dL in women; (b) poor blood pressure control—that is, systolic blood pressure ≥160 or ≤90 mm Hg and/or diastolic blood pressure ≥90 or ≤50 mm Hg caused by either the administration or failure to administer the patient’s regular antihypertensive medication when the route of administration as prescribed was tolerated and the treatment was clinically indicated; (c) poor anticoagulation control—that is, (c.1) international normalised ratio above or below the target therapeutic range; (c.2) pulmonary embolism and venous thromboembolism; (c.3) haematoma in patients with anticoagulation; (c.4) bleeding in patients with anticoagulation; (d) poor control of serum potassium—that is, K+ ≤3.5 mmol/L or K+ ≥5.1 mmol/L; (e) poor heart rate (HR) control—that is, HR ≥120 beats/min or HR ≤45 beats/min at rest in patients receiving treatment with negative chronotropic drugs.
In addition, we recorded the ATC code of the drug involved in the DNO and the negative health outcome caused in the patient.
Patients readmitted within 180 days to the same ED and/or to the hospital ward: patients readmitted owing to decompensation of HF and/or exacerbation of COPD within 180 days after inclusion in the study.
Mean stay: duration, in hours, of the patient’s hospital stay from ED admission to discharge from the ED or the hospital ward.
Mortality at 180 days: this is confirmed through review of the clinical history 180 days after inclusion in the study.
Sample-size calculation
The sample size was calculated considering a statistical power of 80% and a statistical significance level of 5%.
According to the literature, it is likely that there would be potential drug-related problems that needed to be resolved in both the intervention and control groups. It was estimated that the prevalence of resolved potential drug-related problems might be up to 35% and 10% of the patients in the intervention4 and control18 groups, respectively. We assumed that 10% of patients would be lost to follow-up. On the basis of these values, we calculated a sample size of 45 patients was needed in each of the study groups.
Statistical analysis
Frequencies, proportions, means, SD and cross-tabulations were used for the descriptive analysis. t-Tests for independent data where used to compare the mean number of DNO and mean hospital stay between the groups. Chi-square tests were used to compare DNO, admissions/readmissions to the same ED and/or the same hospital and 180-day mortality. All tests were two-tailed and significance was set at p<0.05. We used SPSS version 18 (SPSS Inc, Chicago, IL, USA) for all analyses.
Results
Participant flow
A total of 118 patients were included between 2 January 2012 and 26 February 2013: 59 in each of the intervention and control groups (figure 2). One patient fulfilling the inclusion criteria was not included owing to very poor short-term prognosis. Recruitment ended when the calculated sample was reached. The follow-up period finished on 24 August 2013.
Participant flow ED: emergency department.
Baseline data
Baseline characteristics of patients in the two study groups were similar, except in the case of dyslipidaemia, which was more prevalent in the intervention group (table 1).
Patients’ demographic and baseline clinical characteristics
Outcomes and estimations
The pharmaceutical care programme conducted in the intervention group resolved 82 potential drug-related problems (PDRP) in 43 (72.9%) patients—an average of 1.39 PDRP per patient. Most of these PDRP (n=54, 65.9%) were detected during the medication reconciliation process. More than half of the PDRP (n=49, 59.8%) involved drugs labelled as one of the following three first-level ATC codes: 28 drugs (34.1%) labelled as code C (cardiovascular system), 11 drugs (13.4%) as code B (blood and blood-forming organs) and 10 drugs (12.2%) as code A (alimentary tract and metabolism) (table 2).
Description of the resolved PDRP with the pharmaceutical care programme
Through retrospective analysis of the electronic health record, we detected overall 56 DNO (0.95 per patient) in the intervention group and 85 DNO (1.44 per patient) in the control group (p=0.01). If distributed by patients, DNO were detected in 37 (62.7%) patients in the intervention group versus 47 (79.7%) in the control group (p=0.042). Table 3 summarises the distribution and types of DNO, as well as the ATC first-level code for the drugs involved.
Distribution of DNO in the intervention and control groups
The mean hospital stay in the intervention group was 194.7 hours in the intervention group versus 242.5 hours in the control group (p=0.186). Moreover, 32 (54.2%) patients in the intervention group had been subsequently admitted to hospital throughout the 180 days after being first admitted for HF/COPD decompensation, compared with 22 (37.3%) in the control group (p=0.065).
Mortality at 180 days was detected in 11 (18.6%) patients in the intervention group compared with 13 (22%) patients in the control group (p=0.647).
Discussion
We found that the participants attending the ED with exacerbation of COPD and HF were mostly old, multimorbid adults with polypharmacy. It is particularly noticeable that 80% of the study participants admitted to the ED were later transferred to a hospital ward, while the usual percentage among all-cause admissions is only 10%.19 This result, along with the high risk for DNO seen in the study population,20 reinforces the need to explore the potential benefits of programmes focusing on the resolution of potential drug-related problems in patients presenting to the ED with COPD or HF.
Our findings show that a large number of potential drug-related problems can be resolved by the pharmacist for elderly patients with polypharmacy, as found in previous investigations on this group of patients conducted at other EDs across Spain13 and in areas of care other than the ED as shown by Patterson et al in their review update in 2014.21 The considerable frequency/number of resolved potential drug-related problems obtained through implementation of a pharmaceutical care programme at the ED could be explained by several characteristics inherent to this type of hospital department: high turnover of patients, diversity and complexity of comorbid conditions, non-programmable attendance, 24-hour shifts, frequent prescription of high-risk medications, rapid changes in patient clinical status, high number of interruptions and distractions in a changing environment22 and the selection of patients with COPD and HF since these individuals are known to be at higher risk for DNO. Carrying out a medication reconciliation process after an interview with the patient has been shown to be a key tool for the resolution of potential drug-related problems, accounting for more than half of the resolutions. The prevalence of potential drug-related problems related to medication reconciliation found in our study was similar to the findings reported by Kent et al 23 and de Andrés-Lázaro et al.24
In our study, resolving potential drug-related problems through the implementation of a pharmaceutical care programme led to a remarkable decrease in the prevalence and average number of DNO, resulting in a clinically favourable impact on drug-related negative outcomes. However, the prevalence of DNO observed in the intervention group was still considerable and the resolution of potential drug-related problems did not result in a statistically significant impact on admissions/readmissions, mean stay and 180-day mortality, which may be partly explained by the highly complex needs and poor prognosis of most patients admitted to the ED.
The clinical impact of pharmaceutical care programmes focusing on multimorbid adults with polypharmacy has been previously assessed in areas of care other than the ED. Unfortunately, the evidence is limited and conflicting.
In a pilot randomised controlled trial with 41 patients in the USA, Koehler et al 25 used a composite variable combining hospital readmissions and ED visits to evaluate a pharmaceutical care programme that differed from ours in two minor respects: (1) it included a telephone call to patients 1 week after discharge, and (2) it attended to patients within 24 hours of admission to the hospital, but not patients in the ED. They found a lower rate of hospital readmissions/ED visits within 30 days in the intervention group, but not within 60 days. In a larger study with 674 patients, Leendertse et al 26 evaluated the impact of a pharmaceutical care programme integrated into a multidisciplinary team in a primary care environment in the Netherlands. They found no statistically significant differences in mortality, quality of life or DNO. In Denmark, Olesen et al 27 found no differences in mortality or hospital readmissions between the group of patients visited in their homes by a pharmacist and followed up by telephone for a year, compared with the control group receiving standard care.
Special attention should be paid to the negative outcomes related to the pharmacological treatments detected in our study as specific actions could be developed to deal with these problems hereafter. Poor glycaemic control was the most common problem, accounting for 40% of total DNO in both groups. This result may be explained by the lack of a general protocol for the management of hyperglycaemia at the ED when the study was carried out. Our findings also showed a relevant prevalence of DNO related to poor control of blood pressure and coagulation in the control group, particularly in comparison with the results in the intervention group. There is some evidence to support multidisciplinary pharmaceutical care interventions in primary care to improve blood pressure28 29 and anticoagulation control.30 However, to our knowledge, there are no studies assessing the impact of pharmaceutical care programme interventions on the latter clinical variables in a context similar to ours.
Limitations of the study
This study has some limitations. First, geographical differences in healthcare services, including professional roles, management of patients with chronic disease in primary care, access to certain medications or the operation of the ED itself, may lead to alternative outcomes. However, since our programme included the most common activities conducted by pharmacists at this level, we believe there would not be many differences if applied elsewhere. Second, the control and intervention groups were statistically similar for most variables, except for the prevalence of dyslipidaemia. Dyslipidaemia is a cardiovascular risk factor with an impact on specific health outcomes in the long term but it has no known association with DNO.20 Hence, the relevance of this difference is expected to be negligible. Third, a single pharmacist was responsible for the implementation of the programme and the assessment of results, which might have led to observer bias. However, as the outcomes were detected by retrospective revision of the unmodifiable electronic heath record, this bias would be minimised. Also, use of a single pharmacist in the study might have caused interference at various levels, such as a potentially closer relationship with other professionals at the ED, and better knowledge of the protocols. In contrast, providing this type of care by a team of pharmacists might cause variations owing to different capabilities and different degrees of implementation. Using a team of pharmacists is a feature worth exploring in future studies, although we do not believe that the individual capabilities of hospital pharmacists will have a major effect on the care provided as in Spain they follow a 4-year residency programme that provides a uniform qualified specialisation.
Conclusion
A pharmaceutical care programme focusing on the resolution of potential drug-related problems results in a significant reduction in the prevalence of patients experiencing DNO and also in the average number of DNO per patient. For variables such as patients readmitted within 180 days, mean stay or mortality, the impact of the programme remains unclear. Further research is needed to assess the cost-effectiveness associated with the implementation of such programmes to ensure the most beneficial pharmaceutical care services at the ED.
References
Footnotes
EAHP Statement 4: Clinical Pharmacy Services
Contributors AJ, MAM, MIB. Performed the study: AJ, SH, MP. Analyzed the data: AJ, NG, MAM, MIB, SH, MP, MJF. Wrote the paper: AJ, NG, MAM, MIB. Critically revised the paper and approved the final version to be published: AJ, NG, MAM, MIB, SH, MP, MJF.
Competing interests None declared.
Provenance and peer review Not commissioned; externally peer reviewed. We acknowledge the Societat Catalana de Farmacia Clinica (SCFC) for the Joaquim Bonal award recognition.