Article Text
Abstract
Bar code-assisted medication administration (BCMA) is increasingly being adopted as an additional tool in the prevention of medication administration errors. This literature review summarises the evidence behind the effects of BCMA technology on medication safety. Although most studies show an error-reducing effect of BCMA technology, compliance with the new technology after its implementation and the long-term effects on error reduction are often not assessed. Most importantly, the effect of medication error reduction on patient outcomes is limited.
Statistics from Altmetric.com
Introduction
The medication distribution process is an important source of medication errors. Medication error rates reported in the literature vary widely depending on the methodologies and definitions used. A recent review summarised the prevalence of medication errors as 5.7% of administrations (range 0.038–56.1%, n = 31 studies), 1.07 errors per 100 patient-days (range 0.35–12, n = 9) or 6% of patients hospitalised (range 0.93–24%, n = 7).1 Most errors originate in the medication administration process (median 53%, range 9–90.7%).1 With few barriers to prevent them from occurring, only 2% of medication administration errors are intercepted at the patient bedside.2 Bar code-assisted medication administration (BCMA) is increasingly adopted as an additional barrier in the prevention of medication administration errors. In 2009, 27.9% of hospitals in the USA had implemented BCMA,3 which increased to 50.2% in 2011.4
BCMA technology is developed to improve compliance with checking the ‘five rights’ of medication administration: right patient, right route, right drug, right dose and right time. The right patient is identified by matching the unique bar code on the patient wristband to the patient information in the electronic medication administration record (eMAR). The right drug, right dose, right dosage form and right time are checked by matching the bar code on every unit- or multidose medication to the information in the eMAR. In a 2009 position statement, the American Society of Health-System Pharmacists encouraged health systems to adopt BCMA technology to improve patient safety and the accuracy of medication administration and documentation.5 Most studies evaluating the effect of BCMA on medication administration errors have been conducted in the USA. However, this technology is also used in European countries including Denmark, Italy and the Netherlands6 and, in 2006, the Council of Europe Expert Group on Safe Medication Practices also encouraged the use of electronic systems to improve the safety of medication administration.7 In June 2010 the general assembly of the European Association of Hospital Pharmacists called for the implementation of bar-coded single dose-packed drugs in national and European regulations.8
While BCMA as a tool in the prevention of medication administration errors makes intuitive sense, there is limited evidence demonstrating the effect of this intervention on medication administration errors and patient outcomes. In addition, increased workload is a commonly voiced concern by nursing staff as the use of bar coding technology can potentially result in a longer duration of medication administration. This review of the literature focuses on (1) the effect of BCMA on frequency, type and severity of medication administration errors and (2) the effect of BCMA technology on the duration of the medication administration process.
Methods
Study selection
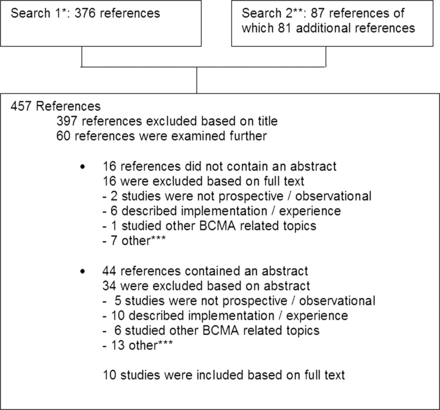
In May 2012 a PubMed search was performed to select studies investigating at least one of the following topics: the effect of BCMA on the rate or severity of medication administration errors or studies evaluating the effect of BCMA on the duration of administering medication. Only studies with a prospective design and in which observational techniques were used to measure medication errors and/or administration time were included. The detailed search criteria and selection procedure of the 10 articles included in this study are shown in figure 1.9–18 We also reviewed the reference lists of the selected articles. This revealed the full text article19 of an abstract we had already selected.15 In addition, we included a study that met the abovementioned criteria and was published in this journal and a Dutch pharmaceutical journal, not indexed in PubMed.20 21 As a result, a total of 11 studies were included.9–14 16–21
Details of literature search. *Search 1: (‘automatic data processing’(MeSH Terms) OR (‘automatic’(All Fields) AND ‘data’(All Fields) AND ‘processing’(All Fields)) OR ‘automatic data processing’(All Fields) OR (‘bar’(All Fields) AND ‘code’(All Fields)) OR ‘bar code’(All Fields) OR ‘barcode’(All Fields)) AND (‘pharmaceutical preparations’(MeSH Terms) OR (‘pharmaceutical’(All Fields) AND ‘preparations’(All Fields)) OR ‘pharmaceutical preparations’(All Fields) OR ‘medication’(All Fields)) AND (‘organisation and administration’(MeSH Terms) OR (‘organisation’(All Fields) AND ‘administration’(All Fields)) OR ‘organisation and administration’(All Fields) OR ‘administration’(All Fields)). **Search 2: (‘pharmaceutical preparations’(MeSH Terms) OR (‘pharmaceutical’(All Fields) AND ‘preparations’(All Fields)) OR ‘pharmaceutical preparations’(All Fields) OR ‘medication’(All Fields)) AND verification(All Fields) AND (‘technology’(MeSH Terms) OR ‘technology’(All Fields)). ***The category ‘other’ includes review articles describing articles on bar code-assisted medication administration (BCMA) and/or technology, summary of a research published in another journal, perspective, editorial, letter to the editor.
Settings and intervention
The studies were conducted on wards with different levels of care and in organisations with varying medication use processes (table 1).
Study characteristics
Implementation of BCMA was accompanied by the implementation of an eMAR in all studies. In two studies the introduction of BCMA was accompanied by additional interventions such as simultaneously implementing bedside assortment picking (table 1).11 20 In all studies, error rates were calculated using the same formula: total errors divided by the sum of observed administrations and omissions. In the study by Franklin et al, bar code technology was used to stock the automated dispensing cabinet and assure the correct identity of the medication. At the bedside, BCMA was then used to assure the correct identity of the patient.11
Results
Error frequency
Error rates before and after implementation of BCMA are summarised in table 2.
Number of observations, and error rates before and after BCMA implementation
As wrong time errors are generally considered to be less severe,22 the results are reported as total errors and errors excluding wrong time errors. Baseline error rate varied between 5.8% and 25.3% if time errors were included and between 1.6% and 27.3% when time errors were excluded. Most studies show a 30–50% reduction in medication administration errors after implementation of BCMA when time errors are excluded. However, implementation of BCMA does not result in a consistent reduction when time errors are included.
Error type
The type and number of error categories varied between studies. Error categories that were assessed in at least three studies and are expected to be reduced by BCMA are omissions, wrong drug errors, unauthorised drug errors, wrong dosage form errors and extra dose errors. Only one study did not find a reduction in unauthorised drug errors and omissions and wrong drug and wrong dose errors even increased.13 Wrong dose errors also increased in the ICU setting in the study by Helmons et al.12 Wrong dosage form errors and extra dose errors increased in the study by Ros et al.20
Wrong route errors are not expected to be influenced by BCMA and wrong time errors only partially. Reduction of these errors was inconsistent among studies. Most studies were underpowered to identify statistically significant differences within individual categories.
Overall it seems that wrong time errors are the most frequently occurring.9 10 13 20
Error severity
Adverse drug events (ADEs) are defined as an injury resulting from the use of a medicine or omission of an intended medicine.23 This definition includes adverse drug reactions and harm from medication incidents. As a result, medication errors resulting in harm are considered ADEs. An error that could potentially lead to harm is a potential ADE. One study14 assessed the severity of observed ADEs and two studies10 11 categorised the potential severity of observed administration errors (table 3).
Severity of observed errors or (potential) ADEs before and after implementation of BCMA
Morris et al found that BCMA reduced the risk of preventable ADEs by 47%14 and Poon et al showed a 50.8% reduction in potential ADEs.10 In this latter study the reduction in many of the potential ADEs could be attributed to improved medication administration documentation.10 Franklin et al did not find a reduction in error severity.11
Duration of medication administration
The general idea that the use of BCMA technology is time-consuming for nursing staff is considered a barrier to implementation. Seven studies addressed this topic (table 4).11 12 16–19 21
Results of studies evaluating the influence of BCMA on time spent on medication administration related tasks
Two studies11 12 evaluated the time spent by nursing staff to complete the medication administration task and three16 17 21 studies measured the duration of each administration. Two studies determined the percentage of total nursing time spent on medication administration by using either the time and motion method15 or the work sampling method.18 No increase in medication administration time was found. Poon et al19 reported a shift in the percentage of time spent on each medication administration task—for example, management of physician orders decreased but verifying patient identity and inefficient waiting increased. Three studies found a reduction in time spent on medication administration.11 16 17
Poon et al19 and Dwibebi et al16 also found that, after implementation of BCMA, the time spent on direct patient care activities increased.
Discussion
The effect of BCMA on the medication error rate is variable among the studies included in this review. BCMA technology seems to decrease the incidence of medication administration errors when time errors are excluded. However, the studies included in this review are heterogeneous.
First, the number and types of administration errors included in the studies vary. In some studies error categories that are not reduced by BCMA are included (eg, technique errors, wrong route errors). This influences the baseline error rate and dilutes the overall effect size of BCMA technology.10–12 21 Second, the study setting has an effect on the baseline prevalence of medication errors and therefore on the potential effect after implementation of BCMA. As an example, medication in an ICU is generally administered intravenously in an area with a higher nurse-to-patient ratio. Indeed, observation of medication administration in an ICU setting resulted in the detection of different types of medication errors than observations performed on a general medicine ward.12 Furthermore, medication use processes varied among the different study settings (table 1)—for example, dispensing of drugs by the pharmacy, use of traditional ward stock or use of automatic dispensing cabinets.
There is also a difference between studies in the time of observation (eg, continuous observation or observing specific medication rounds). As the time of the medication administration round is a determinant for medication errors,24 25 the moment of observation could influence the baseline error rate.
In two studies,11 20 the intervention was comprised of more than BCMA and an eMAR. It is therefore not possible to contribute the error reductions either to BCMA technology or to the other intervention (eg, automated dispensing cabinet) in these studies.
The degree of implementation of the technology is of importance to the results. Shortcomings in design, implementation and workflow integration encourage workarounds.10 19 26 The current study results might therefore reflect the impact of the technology in the context of its implementation rather than the impact of the technology itself.19
Not all studies evaluated user compliance with the new technology. As a result, workarounds could have influenced the effect of BCMA on medication administration errors. Helmons et al and Paoletti et al reported on the compliance rate which was around 90%.9 12 Poon et al reported that 20% of the drugs administered using bar code eMAR technology were given without the bar code scanning step during the study period.10 However, no studies evaluated which errors detected in the study were the result of non-compliance.
Although the goal of BCMA is to enhance medication safety, studies that evaluate the prevention of potential harm after implementation of BCMA are limited.10 11 14 Only two studies showed a reduction in the severity of potential ADEs.14 19 These limited data support the beneficial effects of BCMA and eMAR on patient outcomes.
Evidence on the long-term effect and safety of BCMA is also limited. However, this information is important as workarounds evolve over time. The duration of the positive effects of BCMA on medication administration errors varied from 1 month to 12 months after implementation. Paoletti et al and Poon et al reported data on long-term medication administration error warnings after BCMA implementation. In both studies the number of warnings remained constant during periods of 1.5 and 2 years after implementation of BCMA, respectively, suggesting a long-term effect of this technology in the detection of medication errors.9 27
BCMA did not increase the time spent on medication administration. This is a reassuring finding as nursing staff are concerned about the time-consuming aspects of BCMA technology. The successful implementation of BCMA is the culmination of judicious planning, design, testing, training and support that occurred before, during and after BCMA deployment.19 The degree of implementation of BCMA technology is therefore an important variable in studies evaluating the effect of BCMA.
This review of the literature generally found a positive effect of BCMA on decreasing medication errors without increasing medication administration time. However, these results are difficult to interpret because of the variability in study design, intervention and reporting of outcome measures and confounders. We have created a study design and reporting checklist as a guide for future research in this area (figure 2), although we realise that conducting a study that meets all of these criteria will not be easy.
Checklist for future research on the long-term effect of bar code-assisted medication administration (BCMA) technology on error frequency and severity.
Conclusions
The results of this review generally support the medication administration error reducing potential of BCMA technology up to 1 year after implementation without indications of increasing nursing time spent on medication administration. However, current studies do not always mention user compliance and degree of implementation, factors narrowly related to the effectivity of BCMA technology and necessary to ascertain the maximum achievable effectivity. Future research should focus on the long-term effects of BCMA on medication error reduction, the causes of errors after BCMA implementation, the effects on nursing workflow and the harm prevented by this technology.
References
Footnotes
Contributors JJMH revised the manuscript based on the comments of the reviewers. MMPMJ and PJH thoroughly revised the new version of the manuscript.
Competing interests None.
Provenance and peer review Commissioned; internally peer reviewed.